当科の柏崎大奈が頚動脈プラーク内の低酸素状態がプラーク内の血管新生を誘発して、脳梗塞を引き起こすことを見出し、その論文が米国のJournal of Neurosurgery誌に掲載されました。近い将来、動脈硬化による脳梗塞発症を抑制する創薬のための重要な情報になると考えています。
A Voyage to Depth of Neuroscience Vol. 57
One of our staffs, Dr. Daina Kashiwazaki has published his new article on the mechanism through which ischemic stroke develop due to carotid artery stenosis.
Kashiwazaki D, Koh M, Uchino H, Akioka N, Kuwayama N, Noguchi K, Kuroda S.
J Neurosurg. 2018 Oct 1:1-8
Hypoxia accelerates intraplaque neovascularization derived from endothelial progenitor cells in carotid stenosis.
https://www.ncbi.nlm.nih.gov/pubmed/30485214
Abstract
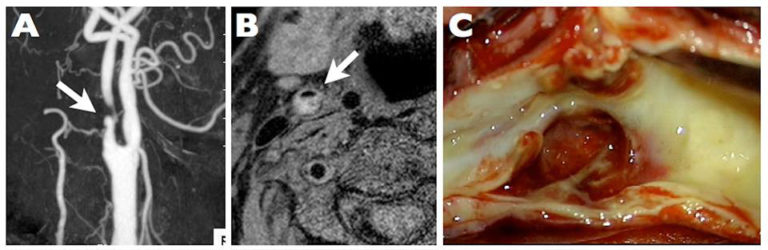
OBJECTIVEThe relationship between intraplaque hypoxia and intraplaque hemorrhage (IPH) has been reported, but the details remain obscure. In this study, the authors aimed to clarify the relationship among intraplaque hypoxia, endothelial progenitor cells (EPCs), and neovascularization, which causes IPH. The histological findings of specimens obtained from carotid endarterectomy were assessed.METHODSThis study included 49 patients who underwent carotid endarterectomy. Magnetic resonance plaque imaging was performed to analyze the components of the carotid plaques, and surgical specimens were subjected to immunohistochemical analysis. The numbers of hypoxia-inducible factor-1 alpha (HIF-1α)-, CD34-, CD133-, and vascular endothelial growth factor receptor-2 (VEGFR-2)-positive cells in the carotid plaques were precisely quantified, as were the number and maximum diameter of CD31-positive microvessels.RESULTSPlaque components were judged as fibrous in 7 samples, lipid-rich in 22, and IPH in 20. The number of CD34-, VEGFR-2-, and CD133-positive cells as an EPC-specific marker was significantly correlated with the number of HIF-1α-positive cells (r = 0.9, r = 0.82, and r = 0.81, respectively). These numbers varied among the 3 plaque components (IPH > lipid-rich > fibrous). The number and maximum luminal diameter of CD31-positive microvessels were also significantly correlated with the number of HIF-1α-positive cells (r = 0.85 and r = 0.89, respectively) and varied among the 3 plaque components (IPH > lipid-rich > fibrous).CONCLUSIONSThe present findings suggest that intraplaque hypoxia may accelerate abnormal microvessel formation derived from EPCs, which in turn promotes IPH. The results also suggest that microvessel enlargement is a pivotal characteristic of IPH and these enlarged microvessels are immature endothelial tubes with disorganized branching and are fragile and prone to rupture.